Projects
Transposable elements in the germline
Transposable elements (TEs) represent ~52% of the human genome and ~40% of the mouse genome, while in both protein coding genes account for only ~1% of the genome. Despite past dismissal of TEs as “selfish genomic elements” or “genomic junk”, their activity controlling organ development and gene expression (acting as cis-regulatory elements) has become apparent. For example, TEs contribute to the derivation of novel genes (including some required for placental function), contribute to 3D genome organization, cis-regulatory elements (including enhancers, promoters, alternative start sites and genomic insulators) and delineate cells which will give rise to the trophectoderm and ICM in the human embryo. While TEs have a roles across mammalian development, I focus on their role during embryonic development of the germline.
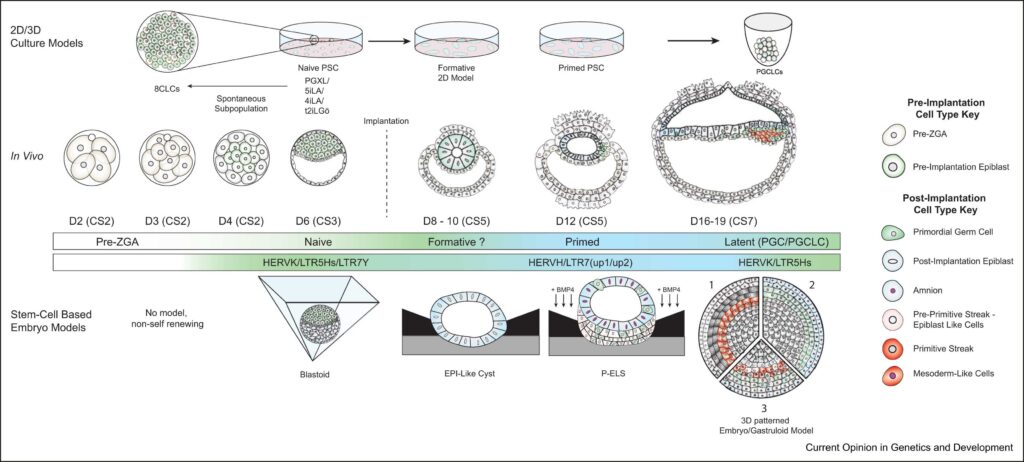
Advances in stem-cell based embryo models allow an opportunity to look for diverse roles of TEs in developing embryonic lineages. Although this work is still in its infancy, you can read my review of TEs, the control of pluripotency, and the promise they offer in understanding embryonic tissues more thoroughly here: DiRusso, J.A and Clark, A.T. Transposable elements in early human embryo development and embryo models. Curr. Opinions in Genetics and Development., August 2023 (Link)
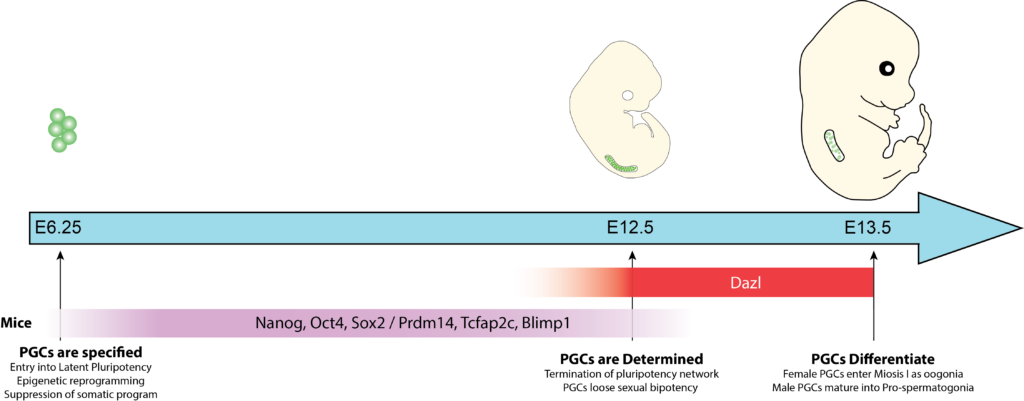
My work in Amander Clark’s lab aims to understand how transposable elements (TEs) are embedded into the epigenetic programming of key fate transitions in mammalian Primordial Germ Cell (PGCs), embryonic precursor to the germline. During development, PGCs undergo two major fate restrictions:
- Specification, in which PGCs are induced in the early embryo, establishing the foundry pool of PGCs.
- Differentiation, in which PGCs become irreversibly committed to the germline and down-regulate key markers of the early PGC program, and enter into sex-specific developmental trajectories
Regulation of Transposable elements is required for gametogenesis
My recent work focuses on understanding how control of TEs in PGCs is required for proper cell fate restriction within the differentiation germline. These transitions occur at a time of extreme epigenetic plasticity, and therefore raise questions about how TEs may be regulated in this window. Insights into this process can teach us how TE control contributes to organismal fitness. This work also demonstrates that TEs are an oft-overlooked aspect in gene regulation and should be considered when deriving in vitro models.
Using an in vivo mouse model, we asked how a master regulator of TEs contributes to proper progression between early PGCs and differentiated germ cells, which lack the early PGC program and are fate-restricted. Using this approach, we find TEs are highly variable in developing PGCs and that their control is gonadal-sex specific. Differences in response to TE misregulation align with known changes in epigenetic control of PGC differentiation, confirming TE control is part of the bona fide PGC program.
More specifics on this project, as well as more information on its findings will be made available when this work is published. Currently, it is under revision in Developmental Cell.
Ltr5Hs in human reproduction
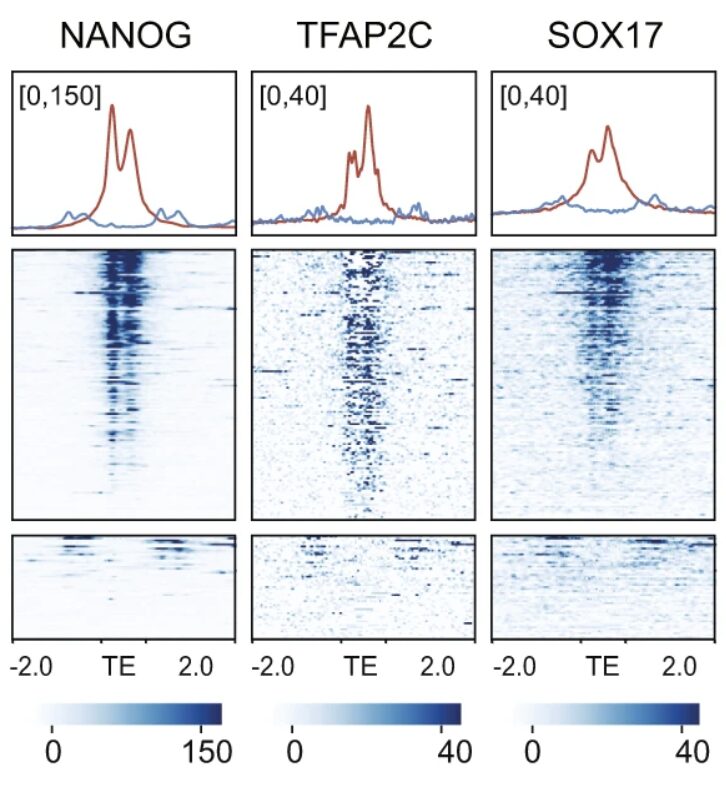
The process by which PGCs arise, called specification, can be modeled in vitro using human pluripotent stem cells. Using directed differentiation of PGC-Like Cells in vitro we found that a young hominidae-specific TE, LTR5Hs, is an essential part of the PGC specification network. My work using this model uncovered extensive binding of SOX17 and NANOG at LTR5Hs elements. Our work demonstrated that TEs have integral roles in the specification of important embryonic lineages, in this case the germline.
This work can be found in here in Nature Communications: Xiang, X.*, Tao, Y.*, DiRusso, J.A., Hsu, F.M., Zhang, J., Xue, Z., Pontis, J., Trono, D., Liu, W., Clark, A.T. Human reproduction is regulated by retrotransposons derived from ancient Hominidae-specific viral infections. Nature Comm., Jan. 2022 *co-first author
Regulation of Organ Homeostasis in Adult Tissues
How organs maintain their size, cell composition, and resume function after wounds or environmental insults remains an open question in cell biology. Using Drosophila as a model, I used high-throughput screening to test how small molecules impacted gut homeostasis and used genetic tools to test their impact on various cell-cell signaling molecules known to function in gut homeostasis.
abcb7 has a drug resistant phenotype
My work on this project showed that ABCB7, an ATP-binding Cassette transmembrane transporter, facilitates multi-drug resistance in Drosophila intestinal stem cells. ABC transporters are highly conserved, and have previously been implicated in facilitating drug resistance in tumors. This work demonstrates a platform on which these compounds can be tested and provides mechanistic insight into multi-drug resistance phenomena.
This work is availible as pre-print on bioRxiv: Dayton, H*, DiRusso, J.A.*, Kolbert, K., Williamson, O., D’Souza, E., Balcuinute, A.,Liu, V., Mao, D., Dettorre, S., Kondo, S., Markstein, M. ABC transporters confer multidrug resistance to Drosophila Intestinal Stem Cells. * co-first authors
methylmercury as an environmental toxin
Methylmercury is a common environmental toxin – most commonly found in seafood as it bio-accumulates in the ocean. It is a particular concern near industrial zones, from which mercury can be in emissions and accumulate in ocean flora and fauna. Because methylmercury is bio-availible, it is also rapidly absorbed from contaminated foods. Methylmercury was identified as an intestinal irritant from a high-throughput luciferase-assay based screen. My work characterized how cell-cell signaling drives the intestinal response to methyl-mercury, showing that irritation is driven though JAK-STAT, rather than JNK signaling.
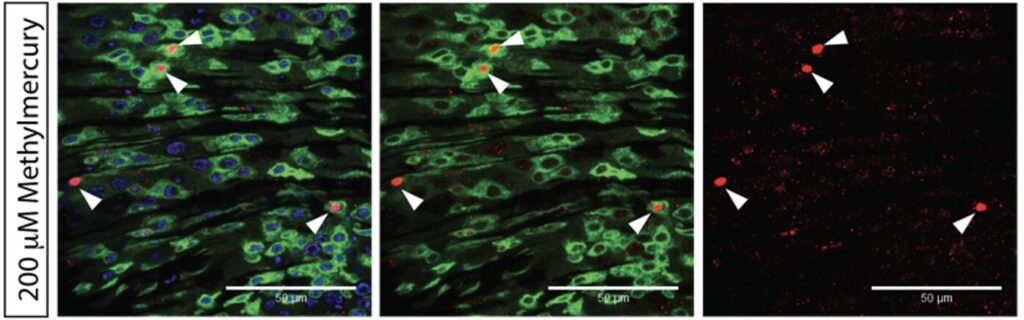
This work was part of my honors thesis at UMass Amherst and can be produced upon request by emailing me here.